Stratasys J5 MediJet 3D Printer
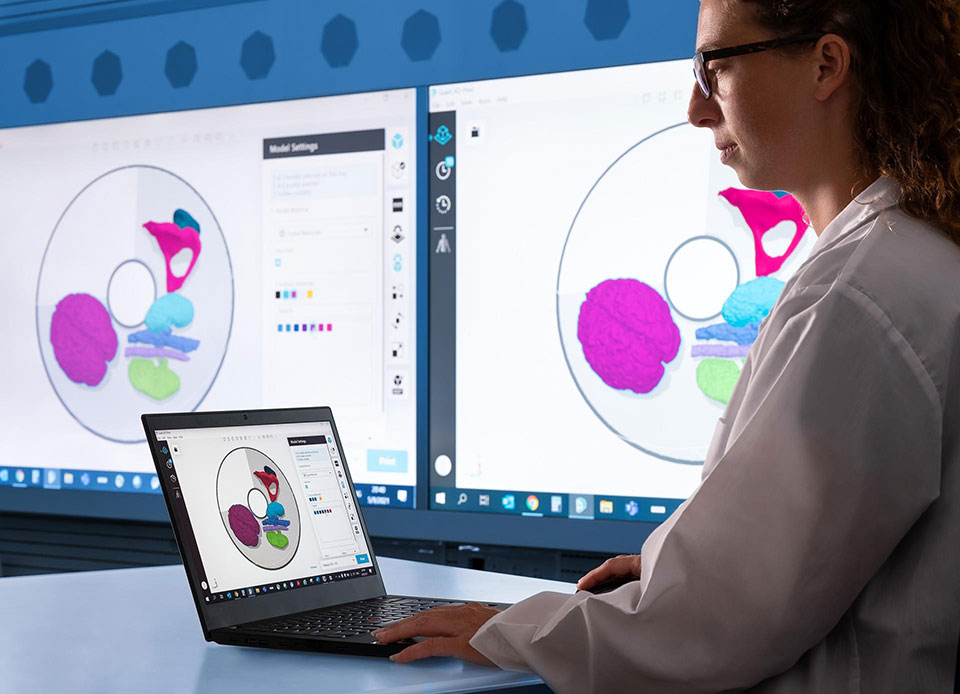
The Stratasys J5 MediJet™ 3D Printer sets a new standard for medical modeling. With multiple materials and multicolor capabilities, academic medical centers, hospitals and medical device companies can create brilliantly vivid pre-surgical planning models, education and training models, medical device development models and drilling and cutting guides* that are sterilizable and biocompatible – all in one platform.
Description
Improve point-of-care planning, patient satisfaction, and training and education.
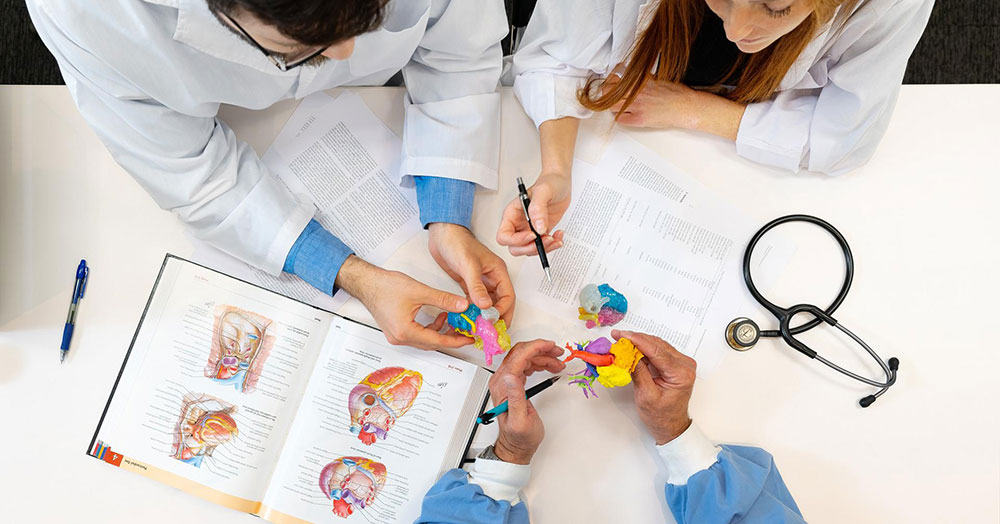
Using patient-specific 3D printed medical models for on-demand training and pre-surgical planning can improve patient outcomes by reducing complications and decreasing OR time and length of hospital stay. Data demonstrates that patient satisfaction improves when 3D medical models are used as part of education for informed consent.
Enhance training and education programs and improve product quality.
Create consistency in new medical device development and testing to enhance product quality, reduce costs and accelerate time to market. Scale up product demonstrations when training field staff and physicians on a new medical device by providing models that replicate the disease state your device is intended to treat—no storage requirements or ethical concerns associated with animal and cadaver models.
All on a certified system.
- 510k cleared for clinical diagnostic use with leading segmentation software companies
- Biocompatibility certification
– ISO 10993-1:2018 for limited contact to tissue and bone and permanent contact to intact skin
– ISO 18562-1:2017 for breathing gas pathways in healthcare applications - Sterilization methods
– Steam, Gamma and EtO for MED610 and MED615RGD
– Steam, Gamma, and EtO for Rigid Transparent family - ISO 13485 Certified (material and hardware manufacturing sites)
Applications
- Patient-specific anatomical models for pre-surgical planning
- Precisely accurate anatomy for training and education
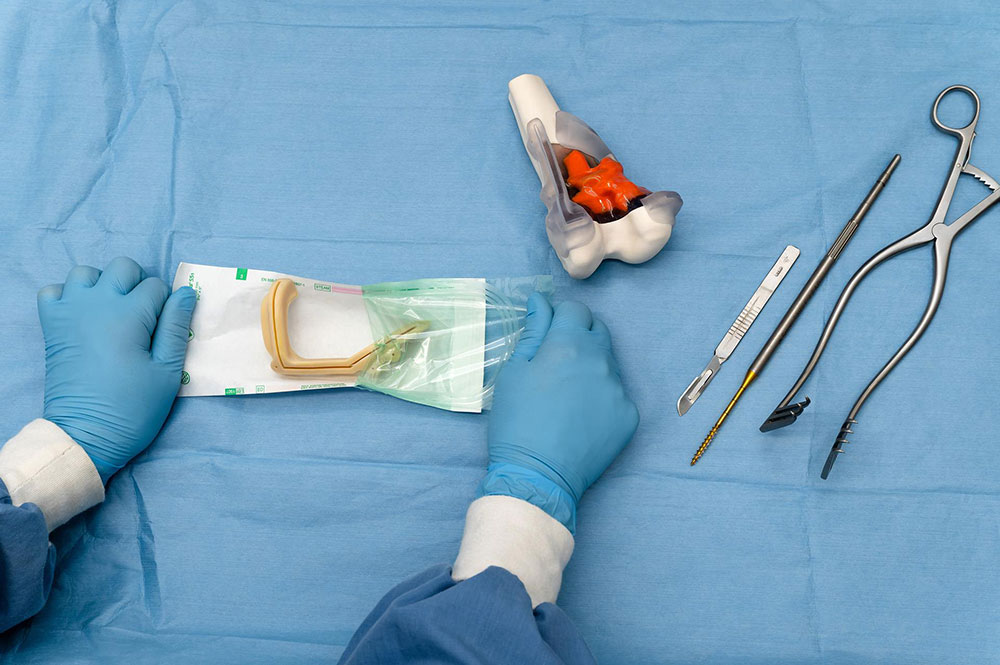
- Surgical guides and tooling*
- Medical device product development
*with approved 3rd party 510k cleared segmentation software
Download the Stratasys J5 MediJet brochure.
Download the Stratasys Medical Modeling Solutions Comparison Chart.
Visit Stratasys for more information.